Welcome to Everlife-Lifeline
South East Asia’s preferred partner for diagnostic and scientific solutions.
Building on a Legacy in Asia
Lifeline Diagnostics Supplies, Inc. is a part of the Everlife Group, Asia’s leading and most trusted distribution platform of medical devices and laboratory equipment and supplies.
As a pan-regional solutions platform for the diagnostic, research and analytical lab markets, Everlife aims to Enable Better Outcomes for the people in our region by improving access to diagnostic and scientific solutions that improve lives. By building a more efficient value chain, new technology can reach the fragmented markets of Asia with greater speed and ease
Our deep understanding of local markets, strong technical expertise and wide commercial networks have helped us become one of the region’s leading solutions platform in 3 major segments:

Clinical Diagnostics

Analytical and Industrial Solutions

Life Sciences
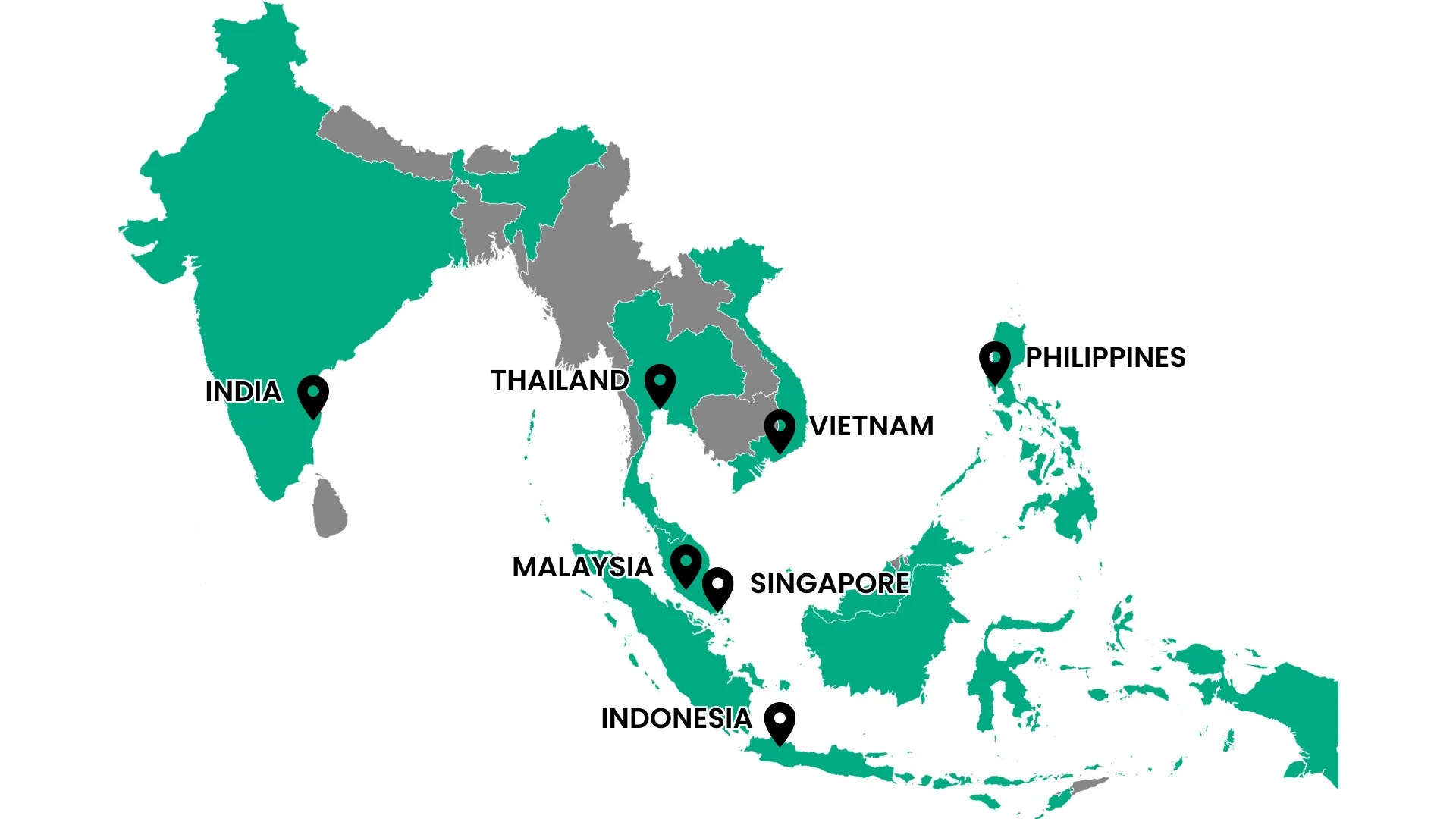
Direct presence in 7 of Asia’s leading markets and expanding
As a full-service partner, we also provide support throughout the value chain and a range of custom solutions, including:
- Regulatory and Licensing
- Importation, Logistics and Warehousing
- Technical Support and Troubleshooting
- Instrument Installation, Maintenance and Servicing
- Design and Manufacturing of Laboratory Furniture
- Clinical and Industrial Lab Information Management Systems (LIMS)
- Turnkey Laboratory Project Management and Consultation
Our Vision
To advance health and quality of life in Asia by improving access to health technology and science.
Our Mission
To improve access to the diagnostic and scientific technology that improves health, quality, environmental and safety outcomes.
Our Values

Aspiration

Agility

Curiosity

External Focus

Collaboration

Integrity
Our values define who we are. By living these values day-by-day, we aim to build a culture that will nurture long-term relationships and bring lasting value to our suppliers, customers, employees and stakeholders. We are committed to:
Providing the best possible products and services to our customers
Building a robust and sustainable business based on strong fundamentals
Cultivating a high-performing, motivated and engaged workforce
Operating under strict compliance rules that govern quality, regulatory, environmental and ethical business practices
Investing in projects and improvements that will benefit the company and community
Contact
Get in Touch
We'd love to hear from you.
Our Address
1225 Quezon Avenue, Quezon City, Philippines 1104
Email Us
info@lifelinediag.com
Call Us
+63 2 8376 5917